Introduction
Psychiatric illnesses affect more than one billion individuals globally and constitute a major public health challenge. Disorders such as major depressive disorder (MDD), schizophrenia, bipolar disorder, anxiety disorders, and postpartum depression (PPD) significantly impair quality of life and productivity while increasing mortality through suicide and comorbid medical conditions. Although pharmacotherapy remains a cornerstone of psychiatric treatment, progress in drug development was relatively stagnant for several decades, relying heavily on monoamine-based antidepressants and dopamine D2 receptor antagonists (1, 2).
Selective serotonin reuptake inhibitors (SSRIs), serotonin–norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants, and classical antipsychotics have undoubtedly improved outcomes for many patients; however, their limitations are increasingly recognized. Delayed therapeutic onset, treatment resistance, metabolic and neurological adverse effects, poor adherence, and lack of efficacy for negative and cognitive symptoms are persistent challenges. Approximately one-third of patients with depression fail to achieve remission with existing therapies, underscoring the need for novel approaches (3, 4).
Recent advances in neuroscience, molecular pharmacology, and translational psychiatry have revitalized the psychiatric drug pipeline. The focus has shifted from monoaminergic modulation toward glutamatergic, GABAergic, cholinergic, inflammatory, and neurosteroid pathways, as well as toward personalized medicine strategies. This review synthesizes major advancements in psychiatric medicines over the past decade and evaluates their clinical and translational significance.
Rapid-acting antidepressants and glutamatergic modulation
Ketamine and esketamine
One of the most significant breakthroughs in psychiatric pharmacotherapy has been the discovery of the rapid antidepressant effects of ketamine, a non-competitive N-methyl-D-aspartate (NMDA) receptor antagonist. Unlike conventional antidepressants that require weeks to exert clinical benefit, ketamine produces symptom relief within hours, even in patients with treatment-resistant depression (TRD) and acute suicidal ideation (5).
The antidepressant effects of ketamine are believed to involve glutamatergic disinhibition, enhanced α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor signaling, activation of mammalian target of rapamycin (mTOR) pathways, and synaptogenesis in the prefrontal cortex. These neuroplastic effects represent a departure from traditional monoamine-centric models of depression.
Esketamine, the S-enantiomer of ketamine, was developed as an intranasal formulation and approved for TRD under controlled administration settings. Clinical trials demonstrated its superiority over placebo when used as an adjunct to oral antidepressants. Esketamine has also shown benefit in rapidly reducing suicidal ideation, although sustained efficacy requires repeated dosing and careful monitoring (6, 7).
Despite their promise, ketamine-based therapies present challenges, including dissociative symptoms, blood pressure elevation, potential for misuse, cost, and limited long-term safety data. Nonetheless, they have reshaped antidepressant research by validating glutamate as a therapeutic target.
Neuroactive steroids and postpartum depression
Brexanolone
Postpartum depression (PPD) is a severe mood disorder affecting women following childbirth and is associated with maternal morbidity, impaired mother–infant bonding, and long-term developmental consequences for children. Conventional antidepressants are often slow to act and inadequately address the neurobiological changes associated with PPD (8, 9).
Brexanolone, an intravenous formulation of allopregnanolone, is a neuroactive steroid that positively modulates GABA_A receptors. It was the first drug specifically approved for PPD. Clinical trials demonstrated rapid and sustained reductions in depressive symptoms following a 60-hour continuous infusion (10).
However, brexanolone administration requires hospitalization and monitoring due to risks of excessive sedation and loss of consciousness, limiting accessibility.
Zuranolone
Zuranolone, an oral neuroactive steroid, represents a major advancement by offering a more practical alternative to brexanolone. Approved in 2023 for PPD, zuranolone produces rapid symptom improvement with a short treatment course. Its mechanism similarly involves enhancement of GABAergic inhibitory neurotransmission.
The success of neuroactive steroids highlights the role of hormonal and GABAergic dysregulation in mood disorders and opens avenues for treating other conditions such as major depression and anxiety disorders (11, 12).
Psychedelic-assisted therapies
Psilocybin and mood disorders
The renewed scientific interest in psychedelics marks a notable shift in psychiatric research. Psilocybin, a serotonergic psychedelic acting primarily at 5-HT2A receptors, has demonstrated antidepressant and anxiolytic effects in controlled clinical settings. Studies suggest that psilocybin induces profound alterations in brain network connectivity, facilitating cognitive and emotional flexibility.
Randomized trials in MDD and end-of-life anxiety have shown significant symptom reductions following one or two supervised dosing sessions combined with psychotherapy. However, results have been heterogeneous, and some large trials have failed to demonstrate superiority over active comparators (13).
Challenges and ethical considerations
Psychedelic therapies are uniquely dependent on psychological context, including preparation, therapeutic support, and integration sessions. Regulatory concerns, long-term safety, risk of psychological distress, and scalability remain unresolved. While promising, psychedelic-assisted therapies require rigorous standardization before routine clinical adoption (14, 15).
Novel antipsychotic mechanisms beyond dopamine D2
TAAR1 agonists
Trace amine-associated receptor 1 (TAAR1) has emerged as a novel target for schizophrenia. TAAR1 modulates dopaminergic and serotonergic neurotransmission indirectly, offering the potential to treat psychosis without direct D2 antagonism.
Ulotaront (SEP-363856), a TAAR1 agonist with additional 5-HT1A activity, demonstrated antipsychotic efficacy in early trials with minimal extrapyramidal and metabolic side effects. Although later-stage trials yielded mixed results, TAAR1 remains a promising target, particularly for addressing negative and cognitive symptoms (16–19).
Muscarinic receptor modulation
Another major breakthrough is the development of muscarinic receptor agonists for schizophrenia. Xanomeline, an M1/M4 muscarinic agonist, showed antipsychotic efficacy but caused intolerable peripheral cholinergic side effects. Its combination with trospium, a peripherally acting anticholinergic, resulted in improved tolerability (KarXT).
Late-stage clinical trials demonstrated significant improvements in positive symptoms, validating muscarinic modulation as a non-dopaminergic antipsychotic strategy (20).
Table 1 summarizes the major recent advances in psychiatric medicines, highlighting novel mechanisms of action, clinical indications, and regulatory status.
Repurposing and multimodal agents
Lumateperone is an atypical antipsychotic with a unique pharmacological profile involving serotonin, dopamine, and glutamate modulation. Initially approved for schizophrenia, it has since gained indications for bipolar depression and adjunctive treatment of MDD (22).
Such repurposing strategies accelerate drug availability while maximizing clinical utility, particularly for disorders with overlapping pathophysiology.
Advances in drug delivery and long-acting injectables
Poor medication adherence is a major contributor to relapse in schizophrenia and bipolar disorder. Long-acting injectable (LAI) antipsychotics address this issue by ensuring sustained drug delivery over weeks to months (23).
Recent meta-analyses indicate that LAIs reduce relapse and hospitalization rates compared with oral therapy, particularly in real-world settings. Innovations in formulation technology have improved tolerability and dosing flexibility, reinforcing LAIs as a key component of modern psychiatric care.
Precision psychiatry and pharmacogenomics
Precision psychiatry aims to tailor treatment based on individual biological, genetic, and clinical characteristics. Pharmacogenomic testing has gained attention for guiding antidepressant selection, particularly genes encoding cytochrome P450 enzymes and serotonin transporters.
Meta-analyses suggest modest but significant improvements in response and remission rates when pharmacogenomic guidance is used. While not yet universally adopted, such approaches represent a step toward personalized treatment (24).
Inflammation-targeted and biomarker-guided therapies
Growing evidence implicates immune and inflammatory mechanisms in subsets of depression and psychosis. Elevated inflammatory markers such as C-reactive protein and interleukins have been associated with treatment resistance.
Anti-inflammatory agents, including cytokine inhibitors and non-steroidal anti-inflammatory drugs, show antidepressant effects primarily in patients with elevated baseline inflammation. This supports a stratified treatment model rather than a one-size-fits-all approach (25).
Structural modifications of GLP-1 and marketed GLP-1 receptor agonists
Figure 1 compares the amino acid sequence of native glucagon-like peptide-1 (GLP-1) with various clinically used GLP-1 receptor agonists and highlights the structural modifications responsible for their improved therapeutic profiles. Native GLP-1 is rapidly inactivated by dipeptidyl peptidase-4 (DPP-4), whereas analogues such as exenatide and lixisenatide contain targeted amino acid substitutions that enhance resistance to enzymatic degradation. Longer-acting agents, including liraglutide and semaglutide, are modified by fatty-acid acylation, which promotes albumin binding and prolongs circulation time. Dulaglutide and albiglutide achieve extended half-lives through conjugation to large protein carriers, such as IgG Fc fragments or albumin. These structural strategies collectively improve stability, extend duration of action, and reduce dosing frequency in clinical use.
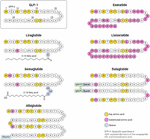
Figure 1. Structural comparison of native GLP-1 and marketed GLP-1 receptor agonists highlighting amino acid substitutions, spacers, and conjugation strategies used to enhance stability and half-life.
Regulatory, safety, and access considerations
Despite scientific progress, access to novel psychiatric medicines remains limited by high costs, regulatory restrictions, infrastructure requirements, and safety concerns. Drugs such as esketamine and brexanolone require supervised administration, increasing healthcare burden.
Post-marketing surveillance and real-world evidence will be critical to defining long-term safety, cost-effectiveness, and optimal patient selection (26).
Future directions
Future psychiatric drug development will likely emphasize:
• Mechanism-based drug discovery beyond monoamines
• Biomarker-driven patient stratification
• Integration of digital and neuroimaging tools
• Combination of pharmacotherapy with psychotherapy and neuromodulation
• Improved accessibility and health-system integration
Conclusion
Recent advances in psychiatric medicines represent a paradigm shift from symptom-based, monoaminergic treatment toward mechanism-driven, rapid-acting, and personalized therapies. Innovations such as glutamatergic antidepressants, neuroactive steroids, novel antipsychotic targets, and precision psychiatry tools offer renewed hope for patients with refractory mental illness. Continued interdisciplinary research, ethical oversight, and health-policy adaptation will be essential to translate these advances into equitable clinical benefit.
Author contributions
Conceptualization, literature search, manuscript drafting, and final approval were performed by the author.
Acknowledgments
We thank the management of Sri Guru Ramdass Group of Institutes, JSS College of Pharmacy, JSS Academy of Higher Education & Research, and PSG College of Pharmacy for their support. Figures were created with BioRender.com. Mendeley Reference Manager assisted in citation management. The authors acknowledge the use of OpenAI to assist in grammar correction, paraphrasing, and improving structural clarity of the manuscript.
Funding
None.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Dardani C, Robinson JW, Jones HJ, Rai D, Stergiakouli E, Grove J, et al. Immunological drivers and potential novel drug targets for major psychiatric, neurodevelopmental, and neurodegenerative conditions. Mol Psychiatry. (2025) 30(10):4487–96. doi: 10.1038/s41380-025-03032-x
2. Paul SM, Potter WZ. Finding new and better treatments for psychiatric disorders. Neuropsychopharmacology. (2024) 49(1):3–9. doi: 10.1038/s41386-023-01690-5
3. Pathak G, Nigade A, Anitha K, Bhatt S. Current drug targets for the treatment of depression. Brain Disord. (2025) 19:100270. doi: 10.1016/j.dscb.2025.100270
4. Zelek-Molik A, Litwa E. Trends in research on novel antidepressant treatments. Front Pharmacol. (2025) 16:1544795. doi: 10.3389/fphar.2025.1544795
5. Popova V, Daly EJ, Trivedi M, Cooper K, Lane R, Lim P, Mazzucco C, et al. Efficacy and Safety of Flexibly Dosed Esketamine Nasal Spray Combined With a Newly Initiated Oral Antidepressant in Treatment-Resistant Depression: A Randomized Double-Blind Active-Controlled Study. Am J Psychiatry. (2019) 176(6):428–38. doi: 10.1176/appi.ajp.2019.19020172
6. Canuso CM, Singh JB, Fedgchin M, Alphs L, Lane R, Lim P, et al. Efficacy and safety of intranasal esketamine for the rapid reduction of symptoms of depression and suicidality in patients at imminent risk for suicide: results of a double-blind, randomized, placebo-controlled study. Am J Psychiatry. (2018) 175(7):620–30. doi: 10.1176/appi.ajp.2018.17060720
7. Wajs E, Aluisio L, Holder R, Daly EJ, Lane R, Lim P, et al. Esketamine nasal spray plus oral antidepressant in patients with treatment-resistant depression. J Clin Psychiatry. (2020) 81(3):19m12891. doi: 10.4088/JCP.19m12891
8. Balthazart J, Ball GF. Endocrine and social regulation of adult neurogenesis in songbirds. Front Neuroendocrinol. (2016) 41:3–22. doi: 10.1016/j.yfrne.2016.03.003
9. O’Donnell KJ, Meaney MJ. Epigenetics, development, and psychopathology. Annu Rev Clin Psychol. (2020) 16(1):327–50. doi: 10.1146/annurev-clinpsy-050718-095530
10. Kanes S, Colquhoun H, Gunduz-Bruce H, Raines S, Arnold R, Schacterle A, et al. Brexanolone (SAGE-547 injection) in post-partum depression: a randomised controlled trial. Lancet. (2017) 390(10093):480–9. doi: 10.1016/S0140-6736(17)31264-3
11. Parikh SV, Aaronson ST, Mathew SJ, Alva G, DeBattista C, Kanes S, et al. Efficacy and safety of zuranolone co-initiated with an antidepressant in adults with major depressive disorder: results from the phase 3 CORAL study. Neuropsychopharmacology. (2024) 49(2):467–75. doi: 10.1038/s41386-023-01751-9
12. Deligiannidis KM, Meltzer-Brody S, Maximos B, Peeper EQ, Freeman M, Lasser R, et al. Zuranolone for the treatment of postpartum depression. Am J Psychiatry. (2023) 180(9):668–75. doi: 10.1176/appi.ajp.20220785
13. Vollenweider FX, Preller KH. Psychedelic drugs: neurobiology and potential for treatment of psychiatric disorders. Nat Rev Neurosci. (2020) 21(11):611–24. doi: 10.1038/s41583-020-0367-2
14. Davis AK, Barrett FS, May DG, Cosimano MP, Sepeda ND, Johnson MW, et al. Effects of psilocybin-assisted therapy on major depressive disorder. JAMA Psychiatry. (2021) 78(5):481. doi: 10.1001/jamapsychiatry.2020.3285
15. Johnson M, Richards W, Griffiths R. Human hallucinogen research: guidelines for safety. J Psychopharmacol. (2008) 22(6):603–20. doi: 10.1177/0269881108093587
16. Berry MD, Gainetdinov RR, Hoener MC, Shahid M. Pharmacology of human trace amine-associated receptors: therapeutic opportunities and challenges. Pharmacol Ther. (2017) 180:161–80. doi: 10.1016/j.pharmthera.2017.07.002
17. Koblan KS, Kent J, Hopkins SC, Krystal JH, Cheng H, Goldman R, et al. A non–D2-receptor-binding drug for the treatment of schizophrenia. N Eng J Med. (2020) 382(16):1497–506. doi: 10.1056/NEJMoa1911772
18. Johnston JN, Kadriu B, Kraus C, Henter ID, Zarate CA. Ketamine in neuropsychiatric disorders: an update. Neuropsychopharmacology. (2024) 49(1):23–40. doi: 10.1038/s41386-023-01632-1
19. Looi JCL, Walterfang M. Striatal morphology as a biomarker in neurodegenerative disease. Mol Psychiatry. (2013) 18(4):417–24. doi: 10.1038/mp.2012.54
20. Simchovitz-Gesher A, Soreq H. Pharmaceutical implications of sex-related RNA divergence in psychiatric disorders. Trends Pharmacol Sci. (2020) 41(11):840–50. doi: 10.1016/j.tips.2020.09.003
21. Brannan SK, Sawchak S, Miller AC, Lieberman JA, Paul SM, Breier A. Muscarinic cholinergic receptor agonist and peripheral antagonist for schizophrenia. N Eng J Med. (2021) 384(8):717–26. doi: 10.1056/NEJMoa2017015
22. Correll CU, Davis RE, Weingart M, Saillard J, O’Gorman C, Kane JM, et al. Efficacy and safety of lumateperone for treatment of schizophrenia. JAMA Psychiatry. (2020) 77(4):349. doi: 10.1001/jamapsychiatry.2019.4379
23. Lahey BB, Moore TM, Kaczkurkin AN, Zald DH. Hierarchical models of psychopathology: empirical support, implications, and remaining issues. World Psychiatry. (2021) 20(1):57–63. doi: 10.1002/wps.20824
24. Insel TR, Cuthbert BN. Brain disorders? Precisely. Science (1979). (2015) 348(6234):499–500. doi: 10.1126/science.aab2358
25. Miller AH, Raison CL. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nat Rev Immunol. (2016) 16(1):22–34. doi: 10.1038/nri.2015.5
26. Treanor M, Rosenberg BM, Craske MG. Pavlovian learning processes in pediatric anxiety disorders: a critical review. Biol Psychiatry. (2021) 89(7):690–6. doi: 10.1016/j.biopsych.2020.09.008
© The Author(s). 2025 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
