Introduction
Breast cancer is a disease with many different types, and each one behaves and reacts to treatment in its own way. New treatments use the molecular markers of each tumor, like estrogen receptor (ER) and human epidermal growth factor receptor 2 (HER2) positivity, PIK3CA and BRCA mutations, neurotrophic tyrosine receptor kinase (NTRK) gene fusions, and other changes that can be targeted, as well as tumor weaknesses like DNA repair problems or the presence of specific antigens. Targeted drugs have saved many lives in metastatic breast cancer (MBC). But many problems are still here, like resistance to treatments, brain metastases, side effects of drugs, and no good options for triple negative breast cancer. This paper looks at new advanced targeted treatments, lists the latest trials and patents from 2023 to 2025, and uses real cases to show how they help people live better (1–3).
Types of Action and Examples of Drugs
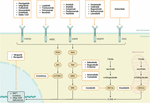
Figure 1 illustrates key oncogenic signaling pathways activated at the cell membrane and how they are pharmacologically targeted by modern precision anticancer therapies. It integrates receptor-level targets, intracellular signaling cascades, metabolic rewiring, and DNA damage repair vulnerabilities.
At the cell membrane, several receptor tyrosine kinases (RTKs) are shown as primary therapeutic targets:
1. FGFR2 (targeted by pemigatinib, infigratinib, futibatinib, erdafitinib, etc.)
2. HER2 (lapatinib, trastuzumab, pertuzumab, neratinib)
3. EGFR (erlotinib, afatinib, cetuximab, panitumumab)
4. VEGFR (vandetanib, cediranib, ramucirumab, lenvatinib)
5. ROS/NTRK signaling (entrectinib)
Activation of these receptors drives major downstream oncogenic pathways:
1. PI3K–AKT–mTOR pathway: This pathway regulates cell growth, metabolism, and survival. Aberrant activation promotes tumor proliferation and resistance to therapy.
(a) Everolimus inhibits mTORC1, suppressing growth signaling.
2. RAS–RAF–MEK–ERK (MAPK) pathway: A central driver of tumor cell proliferation and differentiation.
(a) RAF inhibitors (dabrafenib, regorafenib)
(b) MEK inhibitors (selumetinib, trametinib) block signal propagation.
The diagram also highlights mitochondrial and metabolic reprogramming:
1. IDH1 and IDH2 mutations alter the citric acid cycle, producing the oncometabolite D-2-hydroxyglutarate (D-2HG).
2. Ivosidenib (IDH1) and enasidenib (IDH2) specifically inhibit these mutant enzymes, restoring normal metabolism.
In the nuclear compartment, defects in DNA damage repair (DDR)—including BAP1 mutations—create vulnerabilities exploited by PARP inhibitors such as niraparib and rucaparib, which induce synthetic lethality.
Hormone receptor drugs (ER+ cases)
Endocrine therapy is still the main type for HR+ MBC; recent focused drugs include pills called selective estrogen receptor degraders (SERDs), new selective estrogen receptor modulators (SERMs)/SERDs, and combos with cyclin-dependent kinase 4/6 (CDK4/6) (4, 5) or PI3K inhibitors. New pills like elacestrant and other investigational drugs are being made to fight ER mutations. Triple drug mixes (PI3K + CDK4/6 + fulvestrant) have shown good survival signals in certain HR+ cases with PIK3CA mutations (6, 7).
Cell-cycle targets (CDK4/6)
Palbociclib, ribociclib, and abemaciclib are still the main drugs with hormone therapy. These prevent cancer growth and can extend the time before progression and sometimes overall survival (OS); research continues to see how best to add new drugs or use earlier ones (like with the PATINA trial and others) (8, 9).
Pathway blockers (PI3K/AKT/mTOR)
In some cases, blocking the PIK3CA mutation can help. Drugs like alpelisib and protein kinase B (AKT)/mTOR blockers can benefit certain mutation-carrying patients, but side effects limit their use; newer, more selective, or combo drugs are being tested. Recent triple treatments (like inavolisib + palbociclib + fulvestrant) have shown promising survival advantages in PIK3CA mutants (6, 7, 10–12).
BRCA-targeted drugs (BRCA)
Olaparib and talazoparib are single-ring AER inhibitors that take down tumors with flaws in the main homologous recomb DNA repair job. They cause tumors to die where they have a flaw in breast cancer susceptibility gene ½ (BRCA1/2) jobs. They block the PARP job that fixes single DNA strand breaks. This makes the tumors fall apart when there are two breaks in the DNA that the cancer cell can’t fix because the BRCA isn’t working. These drugs have been tested in patients with a BRCA flaw and shown to help them live longer without the disease showing up. They also make the patients feel better without many bad effects. These drugs work well on the classic BRCA1/2 flaws, but we are also trying to find other flaws that work just as good as BRCA1/2. We are also testing these drugs with other drugs that block the immune system’s stop signs, other drugs that aim at a specific cancer target, or drugs that cause DNA damage. These new ways may help stop tumors that are drug resistant or help more patients benefit from these drugs in a wider group of MBC (13, 14).
Special drugs with antibodies and ADCs
Antibody drug conjugates (ADCs) are a big leap forward for help in cancer of the breast that has spread, a form of treatment that links the precision of common and targeted medicines with the strength of chemo. These new drugs like trastuzumab emtansine T-DM1, trastuzumab deruxtecan, and sacituzumab govitecan are made to send a very strong drug to only the cancer cells that have the right markers on their surface, such as HER2 and Trop 2 (15). The precision of these drugs means they can kill the cancer cells very well while doing less harm to the rest of the body. ADCs have changed how we treat MBC that is HER2 positive and have given new hope to patients with HER2 low and triple negative forms of the disease. These drugs work very well even for patients who have been treated with many drugs before and for those who have stopped responding to chemo, showing they can beat treatment resistance. But these drugs also have different side effects that must be watched closely, including blood problems, stomach issues, and for trastuzumab deruxtecan, a higher chance of lung problems like pneumonitis. Catching and fixing these problems fast is key to giving the best treatment and keeping patients safe (16–21).
Targeted small drugs (NTRK, HER2, etc.)
Tumor-agnostic drugs like the tropomyosin receptor kinase (TRK) inhibitors larotrectinib and entrectinib have shown great and lasting effects on patients with rare NTRK fusion-positive breast cancers, which shows why we should run tests for all genes, even in unusual mutations. At the same time, HER2 treatments are progressing past the simple HER2 overexpression stage, with new tyrosine kinase inhibitors and next-generation antibodydrug combos showing strong effects on HER2 mutant and HER2 low breast cancers in the metastatic form, making targeted drugs available to more people (22, 23).
Immunotherapy and mix-and-match strategies
Drugs blocking the immune system (pembrolizumab, atezolizumab) help in some TNBC cases, especially if the immune system marker programmed death-ligand 1 (PD-L1) is high, combined with chemo. Combining these immune drugs with others (like ADCs or pathway drugs) is being studied to help immune-lacking cases react better to treatment. Ongoing/selected clinical trials (2023–2025)—(Table 1).

Table 1. Below are representative, actively recruiting, or recently reported trials that exemplify modern targeted approaches.
Recent patents and intellectual-property trends (selected examples)
Patent filings emphasize (1) new small-molecule SERDs and ER modulators, (2) ADC linker/cytotoxin optimization and new antigen targets, and (3) combinatorial claims pairing targeted agents with immunotherapy or gene therapies (Table 2).
Selected case studies illustrating targeted therapy impact
Case A — Dramatic response to sacituzumab govitecan in metastatic triple-negative MBC
A published case reported a complete response in a heavily pretreated patient with metastatic metaplastic (TNBC phenotype) breast cancer after sacituzumab govitecan, illustrating ADCs’ capacity to overcome chemotherapy resistance in Trop-2 expressing tumors. This underscores the value of tumor profiling and ADCs in refractory metastatic settings (24).
Case B — NTRK fusion-positive metastatic breast cancer treated with larotrectinib
A case of NTRK3 fusion–positive metastatic secretory carcinoma showed substantial clinical benefit with larotrectinib, supporting tumor-agnostic targeting for rare driver alterations in MBC. Such cases validate broad genomic testing even in breast tumors with unusual histology (25).
Case C — Long-term benefit with T-DM1 biosimilar in HER2+ multisite metastases
Reports of durable disease control using T-DM1 biosimilars in recurrent HER2+ disease demonstrate biosimilars’ potential to expand access while maintaining efficacy but emphasize careful long-term monitoring (3).
Resistance mechanisms and biomarkers
Common barriers include ESR1 mutations, which cause ER therapy failure, and activation of alternative PI3K/AKT pathways, which bypass suppression; in addition, loss of RB1 or amplification of cyclin E leads to resistance to CDK4/6 inhibition, while increased activity of efflux mechanisms and ADC payloads cause drug resistance, and immune escape may hinder checkpoint inhibitor success. Liquid biopsy (ctDNA) is now regularly used to monitor mutation changes over time, such as PIK3CA or ESR1, and new imaging and immune profiling tools are being adopted to help tailor treatments and select drug combinations more precisely. The reports from ASCO/NEJM on triple therapy used ctDNA to detect PIK3CA mutations for patient inclusion, underscoring the key role of molecular markers (26).
Safety, toxicities, and real-world considerations
Targeted agents improve efficacy but have class-specific toxicities: PI3K inhibitors (hyperglycemia, rash), CDK4/6 inhibitors (neutropenia, diarrhea for abemaciclib), ADCs (interstitial lung disease with some HER2-ADCs, severe neutropenia), and PARP inhibitors (hematologic toxicity). Real-world use requires patient selection, monitoring, dose modifications, and multidisciplinary care. Cost and access remain barriers globally (4, 5, 27–30).
Future directions
Key priorities: (1) rational combinations to overcome resistance (e.g., PI3K + CDK4/6 + SERD triple regimens), (2) ADCs with novel antigens/payloads and improved therapeutic indices, (3) tumor-agnostic approaches for rare drivers, (4) integrating immunotherapy with targeted agents to sensitize tumors, (5) expanding genomic and ctDNA-guided adaptive trials (31), and (6) central nervous system (CNS)-penetrant targeted drugs for brain metastases. Patent and trial activity reflect these priorities (32).
Conclusion
Targeted treatments have greatly changed how we treat MBC; choosing the right one (molecular test), flexible treatment plans, and combination approaches now help improve results in certain groups. Still, drug resistance, side effects, access issues, and the need for drugs that work in the brain are key challenges. Many ongoing studies and new patents mean new ideas come fast, often in combinations and with a focus on biomarkers. Doctors should do broad genetic tests and think about putting suitable patients into clinical trials.
List of Abbreviations
MBC: metastatic breast cancer; ER: estrogen receptor; HER2: human epidermal growth factor receptor 2; HR+: hormone receptor–positive; TNBC: triple-negative breast cancer; P13K: phosphatidylinositol 3-kinase; AKT: protein kinase B; mTOR: mammalian target of rapamycin; CDK4/6: cyclin-dependent kinase 4/6; SERDs: selective estrogen receptor degraders; SERMs: selective estrogen receptor modulators; ADCs: antibody–drug conjugates; PARP: poly(ADP-ribose) polymerase; BRCA1/2: breast cancer susceptibility gene 1/2; NTRK: neurotrophic tyrosine receptor kinase; TRK: tropomyosin receptor kinase; PD-L1: programmed death-ligand 1; ctDNA: circulating tumor DNA; PFS: progression-free survival; OS: overall survival; MTD: maximum tolerated dose; CNS: central nervous system.
Author Contributions
Conceptualization, literature search, manuscript drafting, and final approval were performed by the author.
Acknowledgments
We thank the management of Sri Shanmugha College of Pharmacy, JSS College of Pharmacy and PSG College of Pharmacy. Figures were created with BioRender.com. Mendeley Reference Manager assisted in citation management. The authors acknowledge the use of OpenAI, QuillBot to assist in grammar correction, paraphrasing, and improving structural clarity of the manuscript.
Funding
None.
Clinical trial
Not applicable.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Xiong X, Zheng LW, Ding Y, Chen YF, Cai YW, Wang LP , et al. Breast cancer: pathogenesis and treatments. Signal Transduct Target Ther. (2025) 10(1):49. doi: 10.1038/s41392-024-02108-4
2. Nuciforo P, Radosevic-Robin N, Ng T, Scaltriti M. Quantification of HER family receptors in breast cancer. Breast Cancer Res. (2015) 17(1):53. doi: 10.1186/s13058-015-0561-8
3. Yuan Y, Zhou S, Li C, Zhang X, Mao H, Chen W , et al. Cascade downregulation of the HER family by a dual-targeted recombinant protein–drug conjugate to inhibit tumor growth and metastasis. Adv Mater. (2022) 34(23):e2201558. doi: 10.1002/adma.202201558
4. Al-Qasem AJ, Alves CL, Ehmsen S, Tuttolomondo M, Terp MG, Johansen LE , et al. Co-targeting CDK2 and CDK4/6 overcomes resistance to aromatase and CDK4/6 inhibitors in ER+ breast cancer. NPJ Precis Oncol. (2022) 6(1):68. doi: 10.1038/s41698-022-00311-6
5. Glaviano A, Wander SA, Baird RD, Yap KCH, Lam HY, Toi M , et al. Mechanisms of sensitivity and resistance to CDK4/CDK6 inhibitors in hormone receptor-positive breast cancer treatment. Drug Res Updates. (2024) 76:101103. doi: 10.1016/j.drup.2024.101103
6. Bardia A, Cortés J, Bidard FC, Neven P, Garcia-Sáenz J, Aftimos P , et al. Elacestrant in ER+, HER2− metastatic breast cancer with ESR1 -mutated tumors: subgroup analyses from the phase III EMERALD trial by prior duration of endocrine therapy plus CDK4/6 inhibitor and in clinical subgroups. Clin Cancer Res. (2024) 30(19):4299–309. doi: 10.1158/1078-0432.CCR-24-1073
7. Jhaveri KL, Im SA, Saura C, Loibl S, Kalinsky K, Schmid P , et al. Overall Survival with Inavolisib in PIK3CA - mutated advanced breast cancer. N Eng J Med. (2025) 393(2):151–61. doi: 10.1056/NEJMoa2501796
8. Hortobágyi GN, Stemmer SM, Burris HA, Yap YS, Sonke GS, Hart L , et al. Overall survival with ribociclib plus letrozole in advanced breast cancer. N Eng J Med. (2022) 386(10):942–50. doi: 10.1056/NEJMoa2114663
9. Loibl S, Metzger O, Mandrekar SJ, Mundhenke C, Seiler S, Valagussa P , et al. PATINA: a randomized, open label, phase III trial to evaluate the efficacy and safety of palbociclib + Anti-HER2 therapy + endocrine therapy (ET) vs. anti-HER2 therapy + ET after induction treatment for hormone receptor positive (HR+)/HER2-positive metastatic breast cancer (MBC). Ann Oncol. (2018) 29:viii121. doi: 10.1093/annonc/mdy272.357
10. Zhang HP, Jiang RY, Zhu JY, Sun KN, Huang Y, Zhou HH , et al. PI3K/AKT/mTOR signaling pathway: an important driver and therapeutic target in triple-negative breast cancer. Breast Cancer. (2024) 31(4):539–51. doi: 10.1007/s12282-024-01567-5
11. Glaviano A, Foo ASC, Lam HY, Yap KCH, Jacot W, Jones RH , et al. PI3K/AKT/mTOR signaling transduction pathway and targeted therapies in cancer. Mol Cancer. (2023) 22(1):138. doi: 10.1186/s12943-023-01827-6
12. Sharma VR, Gupta GK, Sharma AK, Batra N, Sharma DK, Joshi A , et al. PI3K/Akt/mTOR intracellular pathway and breast cancer: factors, mechanism and regulation. Curr Pharm Des. (2017) 23(11):1633–8. doi: 10.2174/1381612823666161116125218
13. Robson M, Im S-A, Senkus E, Xu B, Domchek SM, Masuda N , et al. Olaparib for metastatic breast cancer in patients with a germline BRCA mutation. N Eng J Med. (2017) 377(6):523–33. doi: 10.1056/NEJMoa1706450
14. Litton JK, Rugo HS, Ettl J, Hurvitz SA, Gonçalves A, Lee KH , et al. Talazoparib in patients with advanced breast cancer and a germline BRCA mutation. N Eng J Med. (2018) 379(8):753–63. doi: 10.1056/NEJMoa1802905
15. Modi S, Jacot W, Yamashita T, Sohn J, Vidal M, Tokunaga E , et al. Trastuzumab deruxtecan in previously treated HER2-low advancedbreast cancer. N Eng J Med. (2022) 387(1):9–20. doi: 10.1056/NEJMoa2203690
16. Crunkhorn S. Increasing stability of ADCs. Nat Rev Drug Discov. (2014) 13(11):812–812. doi: 10.1038/nrd4465
17. Guidi L, Pellizzari G, Tarantino P, Valenza C, Curigliano G. Resistance to antibody-drug conjugates targeting HER2 in breast cancer: molecular landscape and future challenges. Cancers (Basel). (2023) 15(4):1130. doi: 10.3390/cancers15041130
18. Yang T, Li W, Huang T, Zhou J. Antibody-drug conjugates for breast cancer treatment: emerging agents, targets and future directions. Int J Mol Sci. (2023) 24(15):11903. doi: 10.3390/ijms241511903
19. Papavassiliou KA, Sofianidi AA, Papavassiliou AGCAF-. Targeting antibody–drug conjugates (ADCs) in solid cancers. Cancers (Basel). (2025) 17(10):1654. doi: 10.3390/cancers17101654
20. Yajaman DR, Oh Y, Trevino JG, Harrell JC. Advancing antibody–drug conjugates: precision oncology approaches for breast and pancreatic cancers. Cancers (Basel). (2025) 17(11):1792. doi: 10.3390/cancers17111792
21. Valle I, Grinda T, Antonuzzo L, Pistilli B. Antibody–drug conjugates in breast cancer: mechanisms of resistance and future therapeutic perspectives. NPJ Breast Cancer. (2025) 11(1):102. doi: 10.1038/s41523-025-00829-5
22. Subbiah V, Gouda MA, Ryll B, Burris HA, Kurzrock R. The evolving landscape of tissue-agnostic therapies in precision oncology. CA Cancer J Clin. (2024) 74(5):433–52. doi: 10.3322/caac.21844
23. Drilon A, Laetsch TW, Kummar S, DuBois SG, Lassen UN, Demetri GD , et al. Efficacy of larotrectinib in TRK fusion–positive cancers in adults and children. N Eng J Med. (2018) 378(8):731–9. doi: 10.1056/NEJMoa1714448
24. Khan S, Jandrajupalli SB, Bushara NZA, Raja RD, Mirza S, Sharma K , et al. Targeting refractory triple-negative breast cancer with sacituzumab govitecan: a new era in precision medicine. Cells. (2024) 13(24):2126. doi: 10.3390/cells13242126
25. Ando Y, Morita S, Shimokata T, Tsuzuki T, Inafuku S, Iwami K , et al. A rapid and durable response to larotrectinib in a patient with NTRK fusion-positive secretory carcinoma originating from the external auditory canal. Int Cancer Conf J. (2022) 11(4):242–6. doi: 10.1007/s13691-022-00559-6
26. Toy W, Shen Y, Won H, Green B, Sakr RA, Will M , et al. ESR1 ligand-binding domain mutations in hormone-resistant breast cancer. Nat Genet. (2013) 45(12):1439–45. doi: 10.1038/ng.2822
27. Wu Y, Zhang Y, Pi H, Sheng Y. Current therapeutic progress of CDK4/6 inhibitors in breast cancer. Cancer Manag Res. (2020) 12:3477–87. doi: 10.2147/CMAR.S250632
28. Zou Y, Zhang H, Chen P, Tang J, Yang S, Nicot C , et al. Clinical approaches to overcome PARP inhibitor resistance. Mol Cancer. (2025) 24(1):156. doi: 10.1186/s12943-025-02355-1
29. Rios J, Puhalla S. PARP inhibitors in breast cancer: BRCA and beyond. Oncology (Williston Park). (2011) 25(11):1014–25.
30. Swain SM, Shastry M, Hamilton E. Targeting HER2-positive breast cancer: advances and future directions. Nat Rev Drug Discov. (2023) 22(2):101–26. doi: 10.1038/s41573-022-00579-0
31. Zhu X, Chen L, Huang B, Li X, Yang L, Hu X , et al. Efficacy and mechanism of the combination of PARP and CDK4/6 inhibitors in the treatment of triple-negative breast cancer. J Exp Clin Cancer Res. (2021) 40(1):122. doi: 10.1186/s13046-021-01930-w
32. Ding AX, Tang Q, Gao YG, Shi YD, Uzair A, Lu ZL. [12]aneN 3 modified tetraphenylethene molecules as high-performance sensing, condensing, and delivering agents toward DNAs. ACS Appl Mater Interfaces. (2016) 8(23):14367–78. doi: 10.1021/acsami.6b01949
© The Author(s). 2025 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.