Introduction
Historically, pharmaceutics was recognized primarily as a practical science aimed at transforming active pharmaceutical ingredients (APIs) into stable, patient-compliant forms. However, while that remains part of the mission, it is no longer the primary focus or value addition of the profession. The therapeutic landscape has advanced and shifted toward managing large, complex molecules such as nano formulations, nucleic acids, biologics, and highly potent conjugates that require sophisticated formulation and delivery systems to pass through clinical development (1, 2).
Most of the problems associated with poor bioavailability, instability, toxicity at therapeutic doses, and manufacturing difficulties are the main causes of preclinical attrition. These problems are rarely pharmacological; instead, they stem from poor formulation design and process control. That is why, now, pharmaceutics is a key player in the translational medicine landscape, impacting all decisions from candidate choice to regulatory approval (3).
At the same time, improvements in materials science, computer modeling, and manufacturing process analytics have given formulation scientists new tools. Regulatory agencies, understanding the need to modernize, have begun to back innovative manufacturing approaches that focus on quality by design (QbD) and continuous improvement. These changes have made pharmaceutics a field that combines biology, engineering, data science, and regulation (4–6).
Advanced drug delivery systems: focus on lipid nanoparticles
Scientific rationale and formulation principles
Lipid nanoparticles (LNPs) have now become the most proven delivery systems for nucleic acid-based medicines that do not use viruses. They have been successful because the right materials were chosen and not just through testing many different formulae. Typical systems are made of ionizable lipids, phospholipids, cholesterol, and polyethylene glycol (PEG) lipids, and each type of component plays an important functional role (7–9).
Ionizable lipids are the most critical component, as they enable the efficient encapsulation of negatively charged nucleic acids and the escape from endosomes after cells take up the particles. However, minor changes to the structure of these lipids can have a large impact on where the LNPs go in the body, how toxic they are, and how well they transfect cells, illustrating how structure-function relationships in pharmaceuticals can be very sensitive (10).
Clinical translation and ongoing trials
The clinical relevance of LNPs is best illustrated by the growing number of nucleic acid therapeutics currently in human trials.
These programs discussed in Table 1 demonstrate that formulation robustness, rather than biological targeting alone, often determines clinical feasibility. Issues such as immunogenicity, storage stability, and dose reproducibility continue to challenge LNP-based products.
Antibody-drug conjugates: pharmaceutics beyond targeting
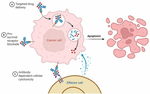
Antibody-drug conjugates (ADCs) were the major example of the union of formulation strategies and biologics, as shown in Figure 1. While target selection is often emphasized when discussing ADCs, the method of assembly critically discusses their efficacy and safety profiles. Achieving the correct chemistry of substance, optimizing drug antibody ratios, preventing particle aggregation, and ensuring formulation stability are essential tasks (11, 12).
Early ADC projects failed because they released the payload too early and weren’t uniform from batch to batch. Now, with advanced site-specific conjugation techniques and better linker technology, reproducibility has gotten better. Still, formulation issues are not easy, especially for low-volume injectable products that require high concentrations (13–15).
Mechanisms of action of antibody-based anticancer therapies
1. Targeted drug delivery: ADCs bind selectively to tumor-associated antigens on cancer cells, are internalized, and release cytotoxic payloads intracellularly, leading to cell death.
2. Pro-survival receptor blockade: Therapeutic antibodies inhibit oncogenic signaling by blocking ligand-receptor interactions or receptor activation on the cancer cell surface, suppressing survival and proliferation pathways.
3. Antibody-dependent cellular cytotoxicity (ADCC): Antibody-coated cancer cells are recognized by Fc receptors on effector immune cells, triggering immune-mediated killing.
Together, these mechanisms culminate in apoptosis and elimination of cancer cells.
These examples discussed in Table 2 reinforce that pharmaceutics governs not only product stability but also therapeutic index and patient safety (16–18).
Artificial intelligence in formulation and process development
In recent years, artificial intelligence (AI) has emerged as a practical tool within the drug development and manufacturing pipeline. Its best uses are in making less work for humans, not in taking their jobs. In the future machine learning will be used more to figure out how a drug dissolves in water, what it looks like, how stable it is, and how it will act (19–23).
In industries, AI and PAT data will be used to control parts of the process to make it better and more accurate, especially in continuous manufacturing (CM). But the powers that be will only let it happen if it can be shown that the AI is clear, proven, and makes sense of the chemistry (24, 25).
Best of all, the AI’s work is only as good as the data it learns from. Most data today is not stored well and is behind a paywall or locked away. This makes a mix of models with data the best way to go (26).
Additive manufacturing and personalized dosage forms
Additive manufacturing now offers new options for patient-specific treatment. Unlike traditional production, 3D printing allows precise control over how the dose looks, how it is structured inside, and how the medication is spread out.
Uses include doses for children, combined pills, and altered-release systems with specific timing. Even with these merits, everyday clinical use is still far off due to lack of clear rules, material challenges, and concern of quality. Pharmaceutics is a major key in setting standards for production that need to be repeatable, robust, and safe for patients in decentralized settings (27).
Continuous manufacturing and regulatory alignment
Continuous manufacturing (CM) is a game changer in the pharma network. It brings the manufacturing process together, allowing immediate monitoring, which makes drug quality more dependable and supply lines safer.
Authorities and organizations have expressed backing for CM through implementing policies that help QbD practices. For pharmaceutics, CM calls for a clearer understanding of process-product relationships and fosters a closer connection between formulation and manufacturing strategies, as shown in Figure 2.
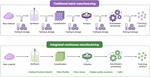
Figure 2. Comparison of traditional batch manufacturing and integrated continuous manufacturing (CM) in pharmaceutical production.
Chart comparing traditional batch manufacturing and CM within the pharmaceutical industry.
The top image shows old-style batch manufacturing: each of these steps—raw material intake, synthesis, crystallization, filtration, downstream processing, and drug finishing—are separate and are followed by many tests and storage periods. This makes the process take longer and use more space, equipment, and energy in the factory. The bottom image shows integrated CM, where these steps are connected in one smooth flow, which can be done live in the factory with less need for storage and intermediate handling. CM has many benefits, including a smaller factory size, more flexible and stable process control, better quality control with live updates, and safer operation.
Patent landscape and intellectual property trends (4, 5)
These patents discussed in Table 3 illustrate a shift from broad platform claims toward highly specific composition and process innovations.
Challenges and future outlook
New ways of making drugs have grown a lot but still have big troubles with how much they costs, who can buy them and all the rules they have worldwide. New ways to bring drugs need clever tech that makes new mixes, better labs, and keeps drugs cold on the way to people who need them most.
Future things we will see will likely come from tools that let us do many things, like mix smarter drugs, make drugs easier to make, and work with rule makers from the start. Encouraging researchers involved in new drug development to work across multiple disciplines within the field will be the most effective way to sustain innovation (23, 28–30).
Conclusion
Pharmaceutics is now a key part of translational medicine. It shows how new therapies are given, made, and checked today. The many new ways to give drugs, use data, and build new ways to make them has made this field larger than the old art of making medicines. When the new ideas are based on real, clinical, and regulatory facts, pharmaceutics will still be a crucial step in turning molecules found in labs into real benefits for patients.
List of abbreviations
ADC: antibody-drug conjugate; AI: artificial intelligence; API: active pharmaceutical ingredient; ATTR: Amyloid transthyretin; CM: continuous manufacturing; CRISPR: clustered regularly interspaced short palindromic repeats; FDA: food and drug administration; HER2: human epidermal growth factor receptor 2; LNP: lipid nanoparticle; mRNA: messenger ribonucleic acid; NSCLC: non–small cell lung cancer; PAT: process analytical technology; PEG: polyethylene glycol; QbD: quality by design
Author Contributions
Conceptualization, literature search, manuscript drafting, and final approval were performed by the author.
Acknowledgments
We thank the management of JKK Munirajah Institute of Health Sciences College of Pharmacy. Figures were created with BioRender.com. Mendeley Reference Manager assisted in citation management. The authors acknowledge the use of OpenAI and QuillBot to assist in grammar correction, paraphrasing, and improving structural clarity of the manuscript.
Funding
None.
Clinical trial
Not applicable.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Uzakova AB, Yergaliyeva EM, Yerlanuly A, Mukatayeva ZS. A systematic review of advanced drug delivery systems: engineering strategies, barrier penetration, and clinical progress (2016–April 2025). Pharmaceutics. (2025) 18(1):11. doi: 10.3390/pharmaceutics18010011
2. Sun X, Setrerrahmane S, Li C, Hu J, Xu H. Nucleic acid drugs: recent progress and future perspectives. Signal Transduct Target Ther. (2024) 9(1):316. doi: 10.1038/s41392-024-02035-4
3. Malamatari M. The importance of drug delivery in the clinical development and lifecycle of drug products with examples from authorised medicinal products. Processes. (2023) 11(10):2919. doi: 10.3390/pr11102919
4. Alemie AA, Siraj EA, Yayehrad AT, Tafere C, Tessema TA, Belete A. Continuous pharmaceutical manufacturing and its contemporary regulatory insights. Disc Appl Sci. (2025) 7(10):1057. doi: 10.1007/s42452-025-07712-9
5. Kashinath KP, Sanjay LR, Ashokbhai MK, Roy S, Sardar MS, Kaity S. Continuous manufacturing based paradigm shift in pharmaceuticals production and current regulatory framework. Chem Eng Res Design. (2025) 215:1–22. doi: 10.1016/j.cherd.2025.01.003
6. Balam R, Mahesh P, Gandhi K, Poojary SG, Chandran A, Vadakkepushpakath AN. Pharma 4.0: enhancing process robustness in pharmaceutical manufacturing through industry 4.0 integration. J Young Pharm. (2025) 17(4):784–9. doi: 10.5530/jyp.20250104
7. Boddu SHS, Vaishya R. Preparation and characterization of folate conjugated nanoparticles of doxorubicin using PLGA-PEG-FOL polymer. Med Chem (Los Angeles). (2012) 02(04):1000117. doi: 10.4172/2161-0444.1000117
8. Hou X, Zaks T, Langer R, Dong Y. Lipid nanoparticles for mRNA delivery. Nat Rev Mater. (2021) 6(12):1078–94. doi: 10.1038/s41578-021-00358-0
9. Cullis PR, Hope MJ. Lipid nanoparticle systems for enabling gene therapies. Mol Ther. (2017) 25(7):1467–75. doi: 10.1016/j.ymthe.2017.03.013
10. Sahay G, Querbes W, Alabi C, Eltoukhy A, Sarkar S, Zurenko C , et al. Efficiency of siRNA delivery by lipid nanoparticles is limited by endocytic recycling. Nat Biotechnol. (2013) 31(7):653–8. doi: 10.1038/nbt.2614
11. Crunkhorn S. Increasing stability of ADCs. Nat Rev Drug Discov. (2014) 13(11):812. doi: 10.1038/nrd4465
12. Zheng H, Zhang Y, Liu L, Wan W, Guo P, Nyström AM , et al. One-pot synthesis of metal–organic frameworks with encapsulated target molecules and their applications for controlled drug delivery. J Am Chem Soc. (2016) 138(3):962–8. doi: 10.1021/jacs.5b11720
13. Yang T, Li W, Huang T, Zhou J. Antibody-drug conjugates for breast cancer treatment: emerging agents, targets and future directions. Int J Mol Sci. (2023) 24(15):11903. doi: 10.3390/ijms241511903
14. Guidi L, Pellizzari G, Tarantino P, Valenza C, Curigliano G. Resistance to antibody-drug conjugates targeting HER2 in breast cancer: molecular landscape and future challenges. Cancers (Basel). (2023) 15(4):1130. doi: 10.3390/cancers15041130
15. Unni A, Ragavendra SP. Engineering T cells for cancer: the evolution and future of CAR-based therapies. Next Res. (2025) 2(4):100892. doi: 10.1016/j.nexres.2025.100892
16. Rojhannezhad M, Soltani BM, Vasei M, Ghorbanmehr N, Mowla SJ. Functional analysis of a putative HER2-associated expressed enhancer, HER2-Enhancer1, in breast cancer cells. Sci Rep. (2023) 13(1):19516. doi: 10.1038/s41598-023-46460-x
17. Swain SM, Shastry M, Hamilton E. Targeting HER2-positive breast cancer: advances and future directions. Nat Rev Drug Discov. (2023) 22(2):101–26. doi: 10.1038/s41573-022-00579-0
18. Khan S, Jandrajupalli SB, Bushara NZA, Raja RDP, Mirza S, Sharma K , et al. Targeting refractory triple-negative breast cancer with Sacituzumab Govitecan: a new era in precision medicine. Cells. (2024) 13(24):2126. doi: 10.3390/cells13242126
19. Garg P, Singhal G, Kulkarni P, Horne D, Salgia R, Singhal SS. Artificial intelligence–driven computational approaches in the development of anticancer drugs. Cancers (Basel). (2024) 16(22):3884. doi: 10.3390/cancers16223884
20. Tousif MN, Akhter R, Ushan MNR, Labonno TM, Mahmud S, Islam Md. N. Revolutionizing cancer therapy: the role of artificial intelligence in enhancing treatment efficacy. In: 2023 International Conference on Information and Communication Technology for Sustainable Development (ICICT4SD). IEEE (2023). p. 89–93. doi: 10.1109/ICICT4SD59951.2023.10303570
21. Singh D, Sachdeva D, Singh L. Advancing breast cancer drug delivery: the transformative potential of bioinformatics and artificial intelligence. Curr Cancer Ther Rev. (2025) 21(3):254–64. doi: 10.2174/0115733947287709240229104857
22. Huhulea EN, Huang L, Eng S, Sumawi B, Huang A, Aifuwa E , et al. Artificial intelligence advancements in oncology: a review of current trends and future directions. Biomedicines. (2025) 13(4):951. doi: 10.3390/biomedicines13040951
23. Nithya R, Venkatesan M, Chandrabose P, Karuppiah Subbulakshmanan NK, Krishnan S. Revolutionizing breast cancer care: the role of artificial intelligence in detection, prediction, and personalized treatment. In: Proceedings of the First International Conference on Science, Engineering and Technology Practices for Sustainable Development, ICSETPSD 2023, 17th-18th November 2023, Coimbatore, Tamilnadu, India. EAI (2024). doi: 10.4108/eai.17-11-2023.2342849
24. Modi S, Jacot W, Yamashita T, Sohn J, Vidal M, Tokunaga E , et al. Trastuzumab deruxtecan in previously treated HER2-low advanced breast cancer. N Engl J Med. (2022) 387(1):9–20. doi: 10.1056/NEJMoa2203690
25. Nami B, Ghanaeian A, Black C, Wang Z. Epigenetic silencing of HER2 expression during epithelial-mesenchymal transition leads to trastuzumab resistance in breast cancer. Life. (2021) 11(9):868. doi: 10.3390/life11090868
26. Albani F, Alghamdi S, Almutairi M, Alqahtani T. Artificial intelligence-driven innovations in oncology drug discovery: transforming traditional pipelines and enhancing drug design. Drug Des Devel Ther. (2025) 19:5685–707. doi: 10.2147/DDDT.S509769
27. Norman J, Madurawe RD, Moore CMV, Khan MA, Khairuzzaman A. A new chapter in pharmaceutical manufacturing: 3D-printed drug products. Adv Drug Deliv Rev. (2017) 108:39–50. doi: 10.1016/j.addr.2016.03.001
28. Wieland M, Fussenegger M. Reprogrammed cell delivery for personalized medicine. Adv Drug Deliv Rev. (2012) 64(13):1477–87. doi: 10.1016/j.addr.2012.06.005
29. Klostermann A, Debertshäuser T, Benary M, Biernath N, Erber B, Tahbaz R , et al. Implementation and outcome of personalized treatment strategies in advanced genitourinary cancers. ESMO Open. (2025) 10(7):105497. doi: 10.1016/j.esmoop.2025.105497
30. Burguin A, Diorio C, Durocher F. Breast cancer treatments: updates and new challenges. J Pers Med. (2021) 11(8):808. doi: 10.3390/jpm11080808
© The Author(s). 2025 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.