Introduction
Cancer continues to be among the leading causes of morbidity and mortality in the world. It is a huge issue even after many years of struggle and development of new methods of treating the disease. Previously, the objective of treating cancer was solely to ensure that the condition disappears permanently. The success was largely determined by the extent to which the tumors were reduced or the duration of time during which the patients remained cancer-free and alive. These are still very important, but they do not give the entire picture of what patients experience (1–3). Our thinking on cancer has evolved in recent years due to various new discoveries in biology, immunology, genetic research, and the way we test new treatments. These changes have been able to save a number of lives and, in some cases, have transformed some of the cancers into conditions that people can live with over a longer period of time. Due to this, the impact of cancer and its cure has become increasingly evident, and a good number of them are reconsidering what true success in curing ought to be like. Nowadays the cancer world understands that to live longer and not to think about whether the patient is well or not in terms of physical, mental, and social levels is not the whole story. Cancer studies have become a major concern now because of the quality of life (QoL) and the success of a patient following cancer treatment. This review examines the ways in which the new science is transforming the duration of life, as well as the QoL, of people. It demonstrates that the objectives of cancer research are evolving in the era of more targeted and customized therapy (4–7).
Evolution of treatment goals in oncology
The initial stage of cancer management was concerned with cytotoxic chemo treatment and big surgeries. Such techniques, though helpful in certain instances, tended to be highly toxic and have long-term health problems. The success was largely determined by scans and rates of survival, and not often by the account of the patient. Combination chemo, adjuvant therapy, and multi-modal treatment began, which increased the rate of cure in certain cancers such as breast cancer, Hodgkin lymphoma, and testicular cancer. However, these benefits were accompanied by some long-term consequences such as cardiovascular damage, secondary cancers, infertility, and brain problems. Consciousness of these effects resulted in a slow increase in the scope of cancer research beyond tumor outcomes. Treatment objectives have become broader in the present day. In addition to controlling tumors, physicians and researchers now consider the feelings of the sick individuals, how many of them work or are able to do their everyday activities, the extent to which they can bear the treatment, and their desires. Regulators and funders have contributed towards this change and now emphasize more on results that are important to patients in clinical trials (8–11).
Advances in immunotherapy and their impact on survival and quality of life
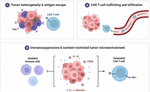
One of the most viable discoveries of the new cancer fight is immunotherapy. Programmed cell death protein 1 (PD-1), programmed cell death ligand 1 (PD-L1), and cytotoxic T lymphocyte antigen 4 immune checkpoint inhibitors have provided long-term outcomes in numerous cancer forms, including melanoma, non-small cell lung cancer, kidney cancer, and head and neck cancer. In contrast to chemo, immunotherapy is able to provide long-term control of the disease in some individuals even after taking medicine. This has brought fresh hope to cancers that had a very bad prognosis. Immunotherapy is typically not as difficult to administer as chemo drugs, in terms of good living, but there are also dangers of immune-related side effects (12–16). Examples of immune-based therapies that can provide more people with an opportunity to live include chimeric antigen receptor (CAR) T-cell therapy (its challenges were shown in Figure 1), bispecific antibodies, and cancer vaccines. However, they may also introduce new issues, including cytokine release syndrome, brain effects, and extremely high costs, potentially disadvantaging patients as well as causing other people to have a difficult time accessing such treatments. Some of the ongoing clinical trials are discussed in Table 1.
This scheme shows the key biological impediments to the effectiveness of CAR T-cell therapy in solid malignancies. (1) Tumor heterogeneity and antigen escape: Solid tumors are made up of different cell types with different or low expression of tumor-associated antigens (TAAs), and this allows cancer cells to avoid CAR T-cell recognition. (2) Damaged CAR T-cell trafficking and infiltration: Tumor vasculature and stromal architecture: Abnormal vascular structure and thick stroma impede effective homing and infiltration of CAR T cells into the tumor mass. (3) Immunosuppressive and metabolically adverse tumor microenvironment: The CAR T-cell dysfunction and exhaustion (inhibitory receptor expression, e.g., PD-1) are caused by inhibitory cytokines (e.g., IL-10, TGF-β) and immunesuppressive cells, hypoxia, and nutrient deprivation (reduced oxygen and glucose). All of these together make CAR T-cell therapy less persistent, active, and antitumor in solid tumors.
Precision oncology and targeted therapies
Precision oncology has revolutionized the process of cancer treatment and concentrated on the molecular fabric of every tumor. Recent technologies such as DNA sequencing and markers have enabled us to identify drugs that are specific to certain cancer-causing genes such as epidermal growth factor receptor (EGFR), human epidermal growth factor receptor 2 (HER2), BRAF, and Kirsten rat sarcoma viral oncogene homolog (KRAS). Therapies targeting these genes have provided improved outcomes and extended lifespans to individuals with numerous types of cancer, and the therapy has a lesser number of side effects than typical chemo (17). This has contributed to improved lives of most patients. Nevertheless, the issue of cancer resistance is massive in scale, and drugs are frequently administered in combinations and observed at the molecular level in patients. Precision oncology has also been aided by liquid biopsy tech to enable us to observe the changes and responses of tumors in real time. Such noninvasive tests imply that patients do not need to receive tissue samples so frequently, and they can detect resistance in the initial stages, which helps patients to treat it much faster and more conveniently (18–24).
Cancer as a chronic disease: implications for survivorship
The number of people who beat cancer is high, and this is a big win for today’s cancer work. But it also brings new issues for the health systems. Survivorship covers the physical, mental, and social effects seen by patients from the time they find out they have cancer until they are out of active treatment for a long time. The long-term survivors may have ongoing symptoms like tiredness, pain, nerve issues, and thinking problems. Also, mental stress, fear of the cancer coming back, and issues fitting back into society are often seen. As survivorship grew, research on it increased, working on ways to help through recovery programs, mental health care, and changes in how people live. Survivorship studies also focus on how important it is to have well-planned follow-up care, which can include watching for delayed effects, other cancers, and other health problems. This full care system shows the new thought that cancer treatment does not end with active care (25–28).
Patient-reported outcomes and quality-of-life assessment
Cancer studies and care now use patient-reported outcome measures (PROMs), which are patient-reported outcome measures. They demonstrate patient attitudes towards symptoms, functioning, and QoL. This provides valuable information that will supplement regular health examinations. The inclusion of PROMs in the studies demonstrates that although treatments may enable patients to live longer, the impacts of the treatment on patient feelings may be quite variable. This has altered the manner in which doctors make decisions in regard to treatments, approval of medicines, and the collaboration of patients and doctors. Evidence on health apps and electronic records also provides further evidence on the QoL among most patient groups. These realities assist in making the treatment of cancer more equal and individualized (29, 30).
Ethical and societal considerations in modern cancer research
Ethical concerns have been compounded with the increase in treatment options. Decisions regarding the intensity of the treatment, whether to continue the treatment in the advanced stage of the disease, and the methods of distributing the limited resources all require careful decisions regarding what is in the best interest of a patient, their QoL, and their preferences. The cost factor has been central in the form of financial strain, particularly expensive targeted medications and immunotherapy. Money trouble may lead to abandonment or worse mental states in patients; hence, the necessity to modify policies and conduct research on the topic of cost and value is high. It is a challenging process to ensure the provision of the best cancer treatments to every corner of the world. The disparities in terms of location, wealth, and health systems continue to influence the outcomes; hence, it is essential to incorporate different studies and collaborate internationally. Some of the recent patents in cancer technologies were discussed in Table 2 (31).

Table 2. Recent patents related to emerging cancer therapies and quality-of-life–focused innovations.
Future directions in cancer research
It is probable that in the future the studies of cancer will combine biological breakthroughs with a patient-oriented perspective. Better detection, better treatment plans, and better survivorship may be achieved by AI, systems biology, and digital health tools. The less strong, yet effective, de-escalation strategies are gaining popularity, particularly in the case of early and low-risk cancers. This is capable of assisting the patients to maintain their life as good without a loss in the length of their life. Finally, it will be the capacity of the modern cancer work to be integrated into new science in the way people live, to ensure that the purpose of life is not only to live longer but also to live better (32–38).
Conclusion
Nowadays research of cancer has passed miles behind attempting to kill tumors. The emergence of new treatments such as immunetherapy, targeted drugs, and new ways of providing care is providing many with a better chance at living, yet it is also highlighting the importance of how people live. With the beginning to treat cancer as a long-term disease, the research should be changed in its direction to survivorship, QoL, and ethics. The human touch is adding to the science of cancer care and is not only prolonging the longevity of people but also their well-being by improving the QoL.
List of abbreviation
CAR: chimeric antigen receptor; CAR-T: chimeric antigen receptor T-cell; TAA: tumor-associated antigen; PD-1: programmed cell death protein 1; PD-L1: programmed cell death ligand 1; CTLA-4: cytotoxic T-lymphocyte–associated protein 4; IL-10: interleukin-10; TGF-β: transforming growth factor-β; ctDNA: circulating tumor DNA, PROMs: patient-reported outcome measures; QoL: quality of life; EGFR: epidermal growth factor receptor; HER2: human epidermal growth factor receptor 2; KRAS: Kirsten rat sarcoma viral oncogene homolog; NSCLC: non-small cell lung cancer; ADC: antibody–drug conjugate; AI: artificial intelligence; APC: antigen-presenting cell; BCMA: B-cell maturation antigen; CDK4/6: cyclin-dependent kinases 4 and 6.
Author contributions
Conceptualization, literature search, manuscript drafting, validation, grammar corrections and final approval were performed by the author.
Acknowledgments
We thank the management of Vellalar College of Pharmacy, Thindal, Erode, Tamil Nadu, India. JSS College of Pharmacy, JSS Academy of Higher Education & Research, Ooty, Nilgiris, Tamil Nadu, India. Lincoln University College, Selangor, Malaysia & SSM College of Pharmacy and United College of Pharmacy, Coimbatore, Tamil Nadu, India. Figures were created with BioRender.com. Mendeley Reference Manager assisted in citation management. The authors acknowledge the use of OpenAI and QuillBot to assist in grammar correction, paraphrasing, and improving structural clarity of the manuscript.
Funding
None.
Clinical trial
Not applicable.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Reya T, Morrison SJ, Clarke MF, Weissman IL. Stem cells, cancer, and cancer stem cells. Nature. (2001) 414(6859):105–11. doi: 10.1038/35102167
2. Wu Y, Zhang Y, Pi H, Sheng Y. Current therapeutic progress of CDK4/6 inhibitors in breast cancer. Cancer Manag Res. (2020) 12:3477–87. doi: 10.2147/CMAR.S250632
3. Gluz O, Liedtke C, Gottschalk N, Pusztai L, Nitz U, Harbeck N. Triple-negative breast cancer—current status and future directions. Ann Oncol. (2009) 20(12):1913–27. doi: 10.1093/annonc/mdp492
4. Gradishar WJ, Anderson BO, Abraham J, Aft R, Agnese D, Allison KH , et al. Breast cancer, version 3.2020, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. (2020) 18(4):452–78. doi: 10.6004/jnccn.2020.0016
5. Yu S, Wang S, Wang X, Xu X. The axis of tumor-associated macrophages, extracellular matrix proteins, and cancer-associated fibroblasts in oncogenesis. Cancer Cell Int. (2024) 24(1):335. doi: 10.1186/s12935-024-03518-8
6. Mukohara T, Park YH, Sommerhalder D, Yonemori K, Kim SB, Kim JH , et al. A phase 1 dose expansion study of a first-in-class KAT6 inhibitor (PF-07248144) in patients with advanced or metastatic ER+ HER2− breast cancer. J Clin Oncol. (2024) 42(16 Suppl):3006. doi: 10.1200/JCO.2024.42.16_suppl.3006
7. Unni A, Ragavendra SP. Engineering T cells for cancer: the evolution and future of CAR-based therapies. Next Res. (2025) 2(4):100892. doi: 10.1016/j.nexres.2025.100892
8. Campbell LL, Polyak K. Breast tumor heterogeneity: cancer stem cells or clonal evolution? Cell Cycle. (2007) 6(19):2332–8. doi: 10.4161/cc.6.19.4914
9. Miller AH, Raison CL. The role of inflammation in depression: from evolutionary imperative to modern treatment target. Nat Rev Immunol. (2016) 16(1):22–34. doi: 10.1038/nri.2015.5
10. Meng Z, Zhang R, Wu X, Zhang M, Jin T. PD-L1 mediates triple-negative breast cancer evolution via the regulation of TAM/M2 polarization. Int J Oncol. (2022) 61(6):150. doi: 10.3892/ijo.2022.5440
11. Hong Y, Boiti A, Vallone D, Foulkes NS. Reactive oxygen species signaling and oxidative stress: transcriptional regulation and evolution. Antioxidants (Basel). (2024) 13(3):312. doi: 10.3390/antiox13030312
12. Jo S, Das S, Williams A, Chretien AS, Pagliardini T, Le Roy A , et al. Endowing universal CAR T-cell with immune-evasive properties using TALEN-gene editing. Nat Commun. (2022) 13(1):3453. doi: 10.1038/s41467-022-30896-2
13. Kundu M, Butti R, Panda VK, Malhotra D, Das S, Mitra T , et al. Modulation of the tumor microenvironment and mechanism of immunotherapy-based drug resistance in breast cancer. Mol Cancer. (2024) 23(1):92. doi: 10.1186/s12943-024-01990-4
14. Santomasso BD, Nastoupil LJ, Adkins S, Lacchetti C, Schneider BJ, Anadkat M , et al. Management of immune-related adverse events in patients treated with chimeric antigen receptor T-cell therapy: ASCO guideline. J Clin Oncol. (2021) 39(35):3978–92. doi: 10.1200/JCO.21.01992
15. Imani S, Farghadani R, Roozitalab G, Maghsoudloo M, Emadi M, Moradi A , et al. Reprogramming the breast tumor immune microenvironment: cold-to-hot transition for enhanced immunotherapy. J Exp Clin Cancer Res. (2025) 44(1):131. doi: 10.1186/s13046-025-03394-8
16. Wu S, Ge A, Deng X, Liu L, Wang Y. Evolving immunotherapeutic solutions for triple-negative breast carcinoma. Cancer Treat Rev. (2024) 130:102817. doi: 10.1016/j.ctrv.2024.102817
17. Zhang J, Xia Y, Zhou X, Yu H, Tan Y, Du Y , et al. Current landscape of personalized clinical treatments for triple-negative breast cancer. Front Pharmacol. (2022) 13:977660. doi: 10.3389/fphar.2022.977660
18. Zhou DD, Bai WQ, Zhai XT, Sun LP, Zhen YS, Li ZR , et al. Excellent effects and possible mechanisms of action of a new antibody–drug conjugate against EGFR-positive triple-negative breast cancer. Mil Med Res. (2021) 8(1):63. doi: 10.1186/s40779-021-00358-9
19. Lin X, Wang G, Liu P, Han L, Wang T, Chen K , et al. Gallic acid suppresses colon cancer proliferation by inhibiting SRC and EGFR phosphorylation. Exp Ther Med. (2021) 21(6):638. doi: 10.3892/etm.2021.10070
20. Widatalla SE, Korolkova OY, Whalen DS, Goodwin JS, Williams KP, Ochieng J , et al. Lapatinib-induced annexin A6 upregulation as an adaptive response of triple-negative breast cancer cells to EGFR tyrosine kinase inhibitors. Carcinogenesis. (2019) 40(8):998–1009. doi: 10.1093/carcin/bgy192
21. Rojhannezhad M, Soltani BM, Vasei M, Ghorbanmehr N, Mowla SJ. Functional analysis of a putative HER2-associated expressed enhancer, Her2-Enhancer1, in breast cancer cells. Sci Rep. (2023) 13(1):19516. doi: 10.1038/s41598-023-46460-x
22. Swain SM, Shastry M, Hamilton E. Targeting HER2-positive breast cancer: advances and future directions. Nat Rev Drug Discov. (2023) 22(2):101–26. doi: 10.1038/s41573-022-00579-0
23. Maruyama K, Shimizu Y, Nomura Y, Oh-hara T, Takahashi Y, Nagayama S , et al. Mechanisms of KRAS inhibitor resistance in KRAS-mutant colorectal cancer harboring HER2 amplification and aberrant KRAS localization. NPJ Precis Oncol. (2025) 9(1):4. doi: 10.1038/s41698-024-00793-6
24. Skoulidis F, Li BT, de Langen AJ, Hong DS, Lena H, Wolf J , et al. Molecular determinants of sotorasib clinical efficacy in KRASG12C-mutated non-small-cell lung cancer. Nat Med. (2025) 31(8):2755–67. doi: 10.1038/s41591-025-03732-5
25. Wagle NS, Nogueira L, Devasia TP, Mariotto AB, Yabroff KR, Islam F , et al. Cancer treatment and survivorship statistics, 2025. CA Cancer J Clin. (2025) 75(4):308–40. doi: 10.3322/caac.70011
26. Lemoine J, Ruella M, Houot R. Born to survive: how cancer cells resist CAR T cell therapy. J Hematol Oncol. (2021) 14(1):199. doi: 10.1186/s13045-021-01209-9
27. Thorsen L, Courneya KS, Steene-Johannessen J, Gran JM, Haugnes HS, Negaard HFS , et al. Association of physical activity with overall mortalityamong long-term testicular cancer survivors: a longitudinal study. Int J Cancer. (2023) 153(8):1512–9. doi: 10.1002/ijc.34625
28. Hussain M, Mateo J, Fizazi K, Saad F, Shore N, Sandhu S , et al. Survival with olaparib in metastatic castration-resistant prostate cancer. N Engl J Med. (2020) 383(24):2345–57. doi: 10.1056/NEJMoa2022485
29. DiPeri TP, Evans KW, Wang B, Zhao M, Akcakanat A, Raso MG , et al. Co-clinical trial of novel bispecific anti-HER2 antibody zanidatamab in patient-derived xenografts. Cancer Discov. (2024) 14(5):828–45. doi: 10.1158/2159-8290.CD-23-0838
30. Berardi R, Morgese F, Rinaldi S, Torniai M, Mentrasti G, Scortichini L , et al. Benefits and limitations of a multidisciplinary approach in cancer patient management. Cancer Manag Res. (2020) 12:9363–74. doi: 10.2147/CMAR.S220976
31. Merker JD, Oxnard GR, Compton C, Diehn M, Hurley P, Lazar AJ , et al. Circulating tumor DNA analysis in patients with cancer: American Society of Clinical Oncology and College of American Pathologists joint review. J Clin Oncol. (2018) 36(16):1631–41. doi: 10.1200/JCO.2017.76.8671
32. Pernas S, Tolaney SM, Winer EP, Goel S. CDK4/6 inhibition in breast cancer: current practice and future directions. Ther Adv Med Oncol. (2018) 10:1758835918786451. doi: 10.1177/1758835918786451
33. Ma L, Guo H, Zhao Y, Liu Z, Wang C, Bu J , et al. Liquid biopsy in cancer: current status, challenges and future prospects. Signal Transduct Target Ther. (2024) 9(1):336. doi: 10.1038/s41392-024-02021-w
34. Valle I, Grinda T, Antonuzzo L, Pistilli B. Antibody–drug conjugates in breast cancer: mechanisms of resistance and future therapeutic perspectives. NPJ Breast Cancer. (2025) 11(1):102. doi: 10.1038/s41523-025-00829-5
35. Garg P, Singhal G, Kulkarni P, Horne D, Salgia R, Singhal SS. Artificial intelligence–driven computational approaches in the development of anticancer drugs. Cancers (Basel). (2024) 16(22):3884. doi: 10.3390/cancers16223884
36. Singh D, Sachdeva D, Singh L. Advancing breast cancer drug delivery: the transformative potential of bioinformatics and artificial intelligence. Curr Cancer Ther Rev. (2025) 21(3):254–64. doi: 10.2174/0115733947287709240229104857
37. Huhulea EN, Huang L, Eng S, Sumawi B, Huang A, Aifuwa E , et al. Artificial intelligence advancements in oncology: a review of current trends and future directions. Biomedicines. (2025) 13(4):951. doi: 10.3390/biomedicines13040951
38. Yu D. Application and prospects of artificial intelligence in breast cancer early diagnosis, screening, and treatment. Trans Mater Biotechnol Life Sci. (2024) 3:213–8. doi: 10.62051/5wdt5q27
© The Author(s). 2025 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.